Intestinal barrier dysfunction — commonly measured through elevated zonulin and lipopolysaccharide (LPS) levels in serum — is increasingly recognized as a driver of systemic inflammation, cognitive degradation, and accelerated biological aging in high-functioning adults. For executives operating under chronic cognitive and physiological load, a compromised gut lining does not remain a localized problem: it becomes a whole-system performance liability, impairing mitochondrial efficiency, disrupting cortisol regulation, and elevating circulating inflammatory cytokines that directly suppress executive function. Targeted probiotic intervention represents one of the most clinically supported strategies for restoring mucosal integrity and recalibrating the inflammatory axis.
The Gut-Brain-Performance Axis: Why Probiotics for Leaky Gut Belong in Your Protocol

The gastrointestinal tract houses approximately 70% of the body's immune tissue. It communicates bidirectionally with the central nervous system through the vagus nerve, the enteric nervous system, and a dense network of neuroendocrine pathways.
This gut-brain axis is not peripheral to executive performance — it is central to it. Dysbiosis, or microbial imbalance, drives elevated interleukin-6 (IL-6) and tumor necrosis factor-alpha (TNF-α). Both cytokines correlate with reduced working memory, impaired decision-making speed, and increased stress reactivity.
Research published in Nature Medicine documented that gut microbiome composition varies significantly between individuals with accelerated versus decelerated biological aging. Microbial diversity emerged as a consistent predictor of systemic inflammaging — the chronic, low-grade inflammatory state underlying most age-related functional decline.
What Leaky Gut Actually Means Clinically — and Why Probiotics Address It

Leaky gut refers to a measurable increase in intestinal permeability. Tight junction proteins between enterocytes break down. This allows bacterial endotoxins such as lipopolysaccharide (LPS) to enter systemic circulation.
Elevated serum LPS activates toll-like receptor 4 (TLR4) signaling, triggering inflammatory cascades that affect liver function, vascular integrity, and neuroinflammation.
Zonulin — a protein regulating tight junctions — is now measurable through serum testing. It serves as a validated biomarker for intestinal permeability. Research published in Gut, a leading gastroenterology journal, has linked elevated zonulin to non-alcoholic fatty liver disease, type 2 diabetes, autoimmune activation, and mood dysregulation. Probiotics for leaky gut work partly by restoring tight junction expression and reducing zonulin output.
The Microbial Architecture Behind a High-Performing Gut
A healthy microbiome is defined by diversity, species richness, and balance among keystone bacterial genera. Lactobacillus and Bifidobacterium species have received the most clinical attention. But Akkermansia muciniphila, Faecalibacterium prausnitzii, and Roseburia intestinalis are equally critical. All three associate with mucosal repair, butyrate production, and anti-inflammatory activity.
Butyrate — a short-chain fatty acid produced through microbial fermentation of dietary fiber — serves as the primary energy source for colonocytes. It directly maintains tight junction integrity.
Populations with depleted butyrate-producing bacteria show measurably higher intestinal permeability. This creates a biochemical basis for using probiotics for leaky gut as a structural intervention, not merely a digestive aid.
Chronic Stress, Cortisol, and Gut Barrier Degradation
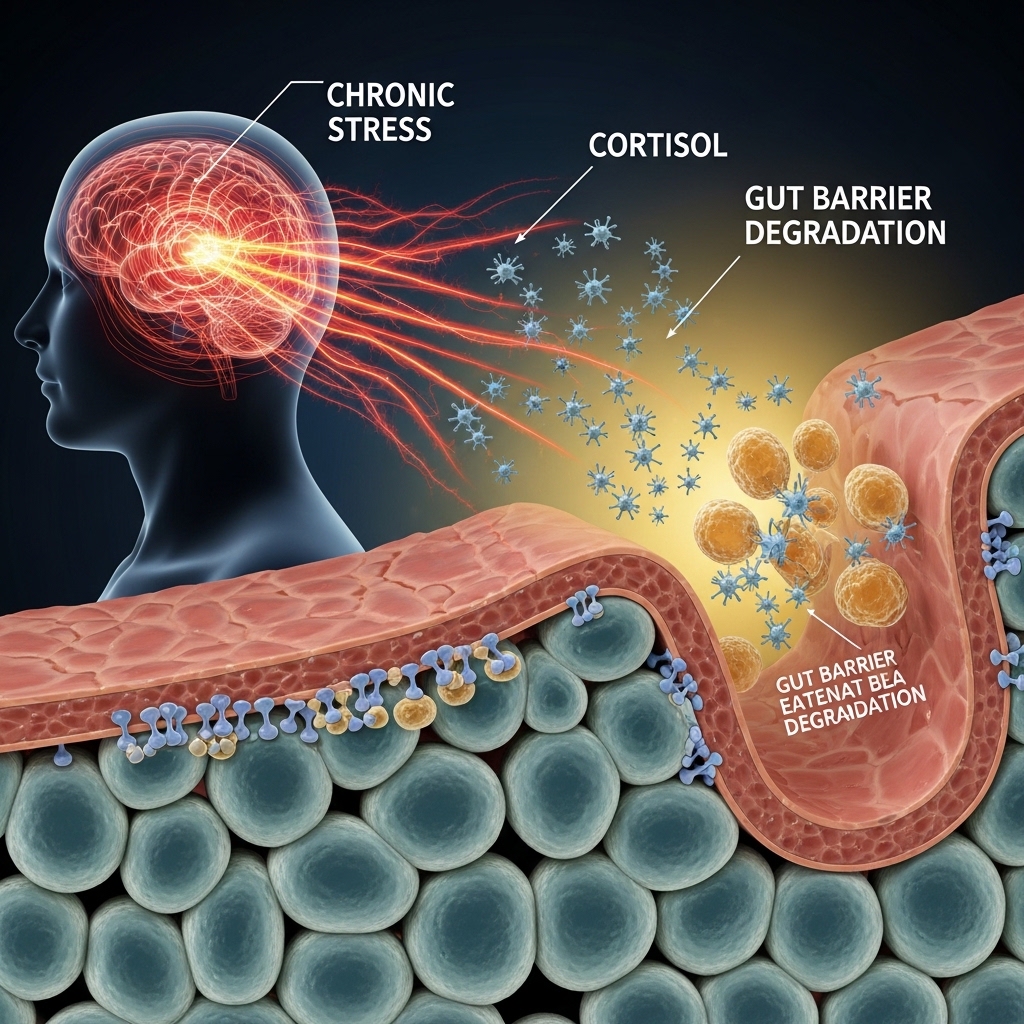
Sustained cortisol elevation activates corticotropin-releasing hormone (CRH) receptors in the gut lining. This increases mast cell degranulation and reduces expression of tight junction proteins — including occludin and claudin-1. The result is a stress-induced increase in intestinal permeability that occurs independently of diet.
A compounding loop then follows. Stress increases permeability. Increased permeability elevates systemic inflammation. Systemic inflammation amplifies stress reactivity through HPA axis sensitization.
Research from the National Institutes of Health documented this bidirectional relationship directly. Gut dysbiosis upregulates HPA axis reactivity — meaning that restoring microbial balance may reduce physiological stress load, not just its downstream effects.
Probiotics for Leaky Gut: Why Strain Selection Determines Clinical Outcome

Not all probiotics operate through the same mechanisms. The clinical evidence is strain-specific. Lactobacillus rhamnosus GG (LGG) ranks among the most studied strains globally. It has documented effects on gut barrier reinforcement and reduction of intestinal permeability markers.
Lactobacillus plantarum 299v has shown efficacy in clinical trials for reducing intestinal inflammation and modulating immune activation in the gut epithelium. Bifidobacterium longum and Bifidobacterium infantis have been studied in the context of stress physiology and neuroinflammation.
Evidence suggests these strains reduce cortisol awakening response and improve psychological resilience in healthy adults under high cognitive load. Generic probiotic products selected on colony-forming unit count alone frequently ignore this strain-level specificity — a clinically significant oversight.
Inflammatory Load, CRP, and the Cardiovascular Risk Connection
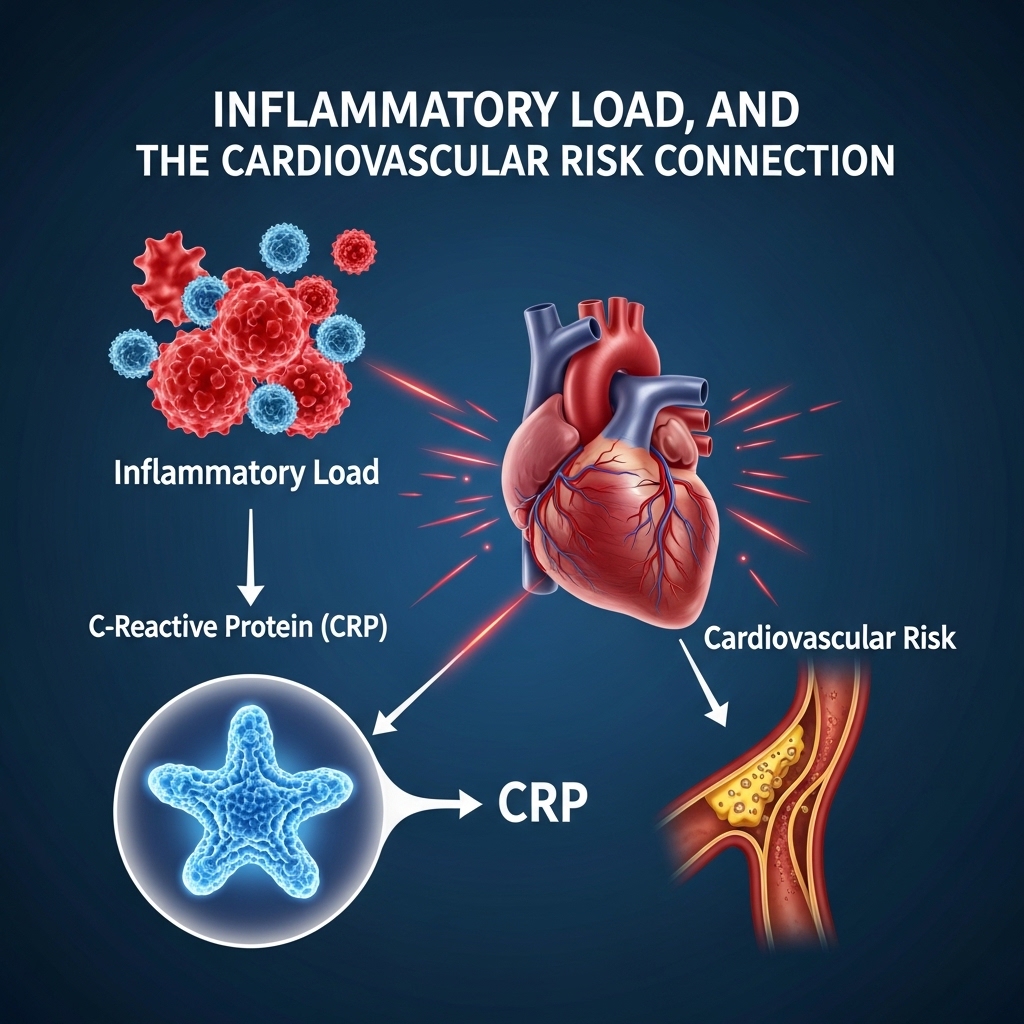
Systemic inflammation from gut barrier dysfunction does not stay confined to the gastrointestinal tract. Circulating LPS and bacterial metabolites drive elevations in high-sensitivity C-reactive protein (hs-CRP) — a cardiovascular risk biomarker tracked in executive health panels worldwide.
The American Heart Association formally recognizes hs-CRP as an independent predictor of cardiovascular events.Multiple randomized controlled trials demonstrate that targeted probiotics for leaky gut reduce hs-CRP in adults with elevated baseline inflammatory markers.
The mechanisms include reduced LPS translocation, improved regulatory T-cell activity, and downregulation of NF-κB — the master transcription factor governing inflammatory gene expression. Executives who monitor cardiovascular biomarkers but overlook gut-origin inflammation face a meaningful diagnostic gap.
READ ALSO: 3 Gut-Friendly Spring Smoothies to Sip This Season
Cognitive Performance and the Psychobiotic Evidence Base

The field of psychobiotics studies probiotics specifically for their effects on brain function and mood. Research from APC Microbiome Ireland, published in peer-reviewed journals including Nutritional Neuroscience, documented that specific probiotic strains reduce perceived stress, lower cortisol output, and improve sustained attention in healthy adults.
The gut produces approximately 90% of the body's serotonin. It also produces significant quantities of gamma-aminobutyric acid (GABA). Gut-derived serotonin does not cross the blood-brain barrier directly.
However, it influences brain signaling through enterochromaffin cells and the vagal nerve pathway. The downstream effects include mood regulation, sleep latency, and emotional reactivity — all of which affect executive decision-making quality.
Metabolic Function: How Probiotics for Leaky Gut Influence Insulin Sensitivity

LPS-driven inflammation impairs insulin receptor signaling. Multiple metabolic research cohorts have documented this mechanism. It contributes to insulin resistance independent of caloric intake or adiposity.
For professionals aged 40 to 60, subclinical metabolic dysfunction often precedes clinical diagnosis by years. Elevated LPS may be a contributing variable in cases where standard dietary explanations fall short.
Research from the Harvard T.H. Chan School of Public Health documented associations between gut microbiome composition and insulin sensitivity. Microbial diversity correlates positively with metabolic flexibility. Probiotic intervention studies targeting metabolic outcomes have shown reductions in fasting glucose, HOMA-IR, and triglyceride levels in adults with dysbiosis-associated metabolic dysfunction.
READ ALSO: 10 Signs of an Unhealthy Gut You Shouldn’t Ignore
Sleep Architecture and the Gut's Circadian Repair Window
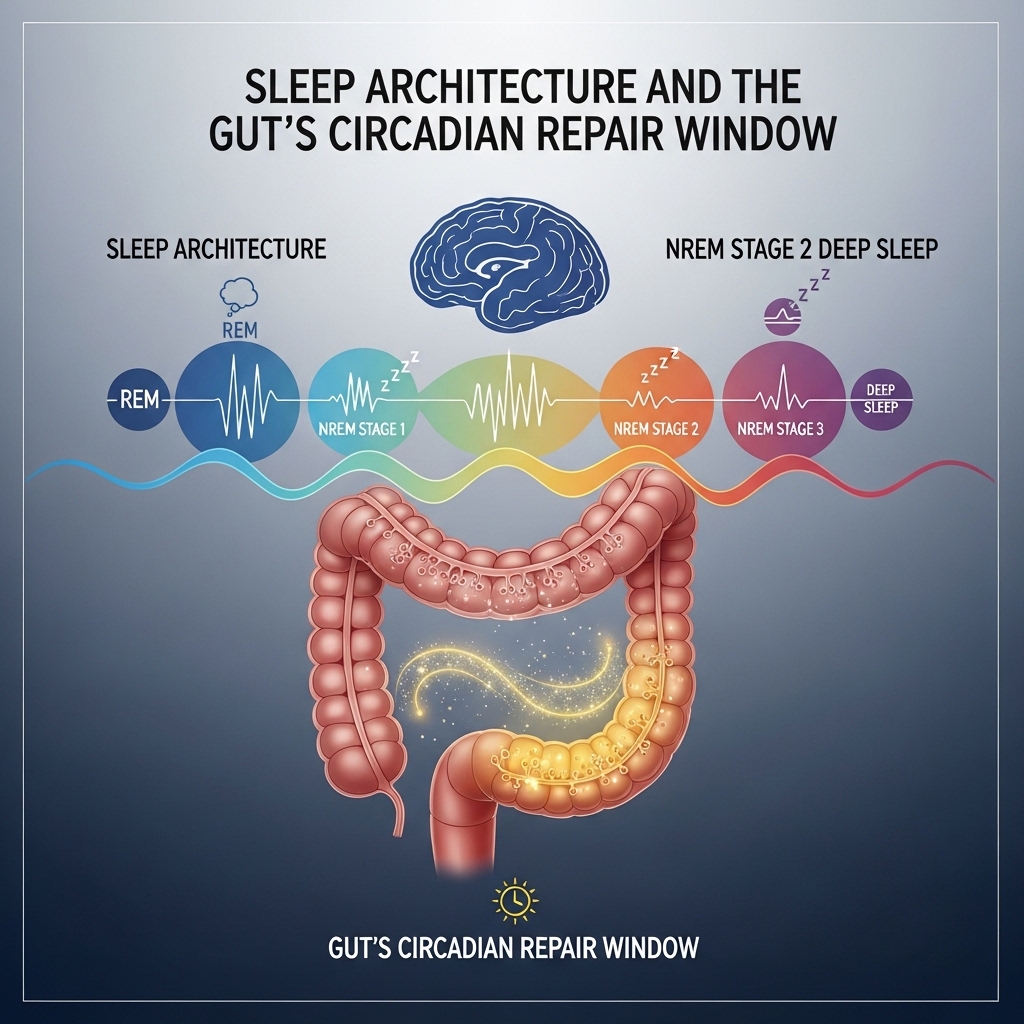
The gut microbiome follows its own circadian rhythm. Microbial activity and tight junction integrity both vary across the 24-hour cycle. Sleep deprivation disrupts intestinal circadian gene expression. It reduces tight junction protein expression during overnight repair windows. It also alters microbial composition within days of sleep restriction onset.
Gut bacteria influence tryptophan bioavailability — a precursor to both serotonin and melatonin. Certain Lactobacillus strains modulate the kynurenine-to-serotonin ratio, a metabolic variable associated with sleep quality and mood stability.
For executives experiencing non-restorative sleep without clear lifestyle explanations, gut-mediated tryptophan dysregulation warrants clinical investigation.
READ ALSO: Gut Cleanse Tips for a Happier Stomach
Dietary Cofactors: What Probiotics for Leaky Gut Cannot Do Alone

Probiotic intervention operates within a broader nutritional context. Probiotics for leaky gut are most effective when paired with adequate prebiotic substrate — fermentable fibers that feed and sustain commensal bacterial populations. Without sufficient dietary fiber from inulin-rich vegetables, legumes, and resistant starches, introduced probiotic strains face a hostile gut environment. Colonization and sustained benefit become unlikely.
Polyphenols — concentrated in berries, olive oil, dark cacao, and green tea — exert documented prebiotic and anti-inflammatory effects on the gut epithelium. They upregulate tight junction protein expression.
Chronic alcohol consumption, ultra-processed food intake, and NSAID use remain among the most documented drivers of gut barrier degradation. Each operates through distinct but overlapping mechanisms that probiotic supplementation alone cannot reverse.
Leaky Gut Testing: Establishing a Baseline Before Intervening

Before selecting a probiotic protocol, baseline assessment clarifies whether intestinal permeability is an active variable in one's current health profile. Serum zonulin and hs-CRP offer accessible starting points. Fasting LPS panels, where available, provide more direct evidence of bacterial translocation.
Comprehensive gut microbiome analysis through validated stool sequencing platforms identifies specific dysbiosis patterns that generic interventions may miss. This diagnostic step matters because gut barrier dysfunction does not always produce obvious gastrointestinal symptoms.
In high-performing professionals, the presenting signs are more likely to be cognitive — brain fog, attentional instability, emotional reactivity under pressure — or metabolic, such as unexplained insulin resistance or persistent inflammation on bloodwork. Identifying the gut as the source requires deliberate testing, not assumption.
READ ALSO: Yeast Nutrition: A Quiet Gut Reset for the New Season
Evidence-Based Next Steps for the High-Performing Professional

The evidence supports a sequenced approach. First, assess baseline markers — serum zonulin, hs-CRP, and microbiome composition — to confirm gut barrier dysfunction as an active variable. Second, consider a multi-strain probiotic formulation with documented clinical backing for barrier repair, not a generic consumer product chosen on colony-forming unit count. Third, increase diverse dietary fiber intake to sustain any introduced strains. Fourth, address the primary gut disruptors — chronic sleep restriction, sustained NSAID use, and ultra-processed food consumption — that supplementation cannot overcome in isolation. Working with a physician who incorporates functional or precision medicine markers, rather than relying on standard metabolic panels, is the most direct path to understanding whether this axis is affecting cognitive output, metabolic function, or inflammatory load in a clinically measurable way.
UP NEXT: Natural Superfoods for Energy: Fuel Your Day the Right Way
Gut barrier dysfunction — measured through elevated zonulin and circulating LPS — drives systemic inflammaging, a process that research associates with accelerated biological age independent of chronological age, lifestyle, or genetic risk factors. WholeLiving's Biological Age Estimation Model incorporates this factor directly — your assessment takes under five minutes.
Ready to understand how these factors are influencing your biological age right now? [Take the Biological Age Assessment →]






